Complete listing on Google Scholar (most publications prior to 2009 are not listed below)
2022
Together, not separately, OH and O3 oxidize Hg (0) to Hg (II) in the atmosphere. P. J. Castro, V. Kellö, I. Cernušák, and T. S. Dibble. J. Phys. Chem. A 2022, 126, 8266–8279.
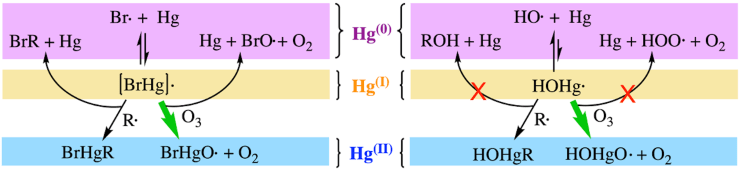
Combined Experimental and Computational Kinetics Studies for the Atmospherically Important BrHg Radical Reacting with NO and O2. R. Wu, P. J. Castro, C. Gaito, K. Beiter, T. S. Dibble, and C. Wang. J. Phys. Chem. A 2022, 126, 3914–3925.
Reaction mechanism and kinetics of the important but neglected reaction Hg with NO2 at low temperature. X. Li, M. Yan, T. S. Dibble, and L. Zhang Chem. Eng. J. 2022, 432, 134373.
Modeling electron beam irradiation of methane. W. Gu and T. S. Dibble, Int. J. Chem. Kinetics 2022, 54, 256-265.
2021
Improved mechanistic model of the atmospheric redox chemistry of mercury. V. Shah et al. Environ. Sci. Technol., 2021, 55. [In which we contradict one of the major points of our 2020 paper on OH-initiated oxidation of Hg(0)! See the Figure below for a summary of the mass flows of Hg.]

Theoretical Study of the Monohydration of Mercury Compounds of Atmospheric Interest. Sonia Taamalli, Michal Pitoňák, Theodore S. Dibble, Ivan Černušák, and Florent Louis. J. Phys. Chem. A 2021, 125, 5819–5828.
2020
BrHgO• + CO: Analog of OH + CO and reduction path for Hg(II) in the atmosphere. D. Khiri, F. Louis, I. Černušák. and T. S. Dibble. ACS Earth and Space Chem., 2020, 10, 1777-1784. [BrHgO radical behaves very, very similarly to OH radical in all its reactions studied to date. Differences in bond energies mean that the BrHgO + CO never gets trapped in a well of BrHgOCO, so there is no pressure dependence of the overall rate constant for making BrHg + CO2.]
First experimental kinetic study of the atmospherically important reaction of BrHg• + NO2. R. Wu, C. Wang. and T. S. Dibble, Chem. Phys. Lett., 2020, 759, 137928. [In which experiment tells us that our previous computations performed rather poorly for getting the low-pressure limiting rate constant; however, experiments tend to confirm the existence of the barrierless reduction reaction predicted by computations.]
Chemical Kinetics and Photochemical Data for Use in Atmospheric Studies, Evaluation No. 19. J. B. Burkholder, S. P. Sander, J. Abbatt, J. R. Barker, C. Cappa, J. D. Crounse, T. S. Dibble, R. E. Huie, C. E. Kolb, M. J. Kurylo, V. L. Orkin, C. J. Percival, D. M. Wilmouth, and P. H. Wine JPL Publication 19-5, Jet Propulsion Laboratory, Pasadena, 2019. http://jpldataeval.jpl.nasa.gov (published July 2020).
Modeling the OH-Initiated Oxidation of Mercury in the Global Atmosphere Without Violating Physical Laws, T. S. Dibble, H. L. Tetu, Y. Jiao, C. P. Thackray, and D. J. Jacob, J. Phys. Chem. A, 2020, 124, 444-453.
In which we:
a) get a very accurate HO-Hg bond energy for HOHg, and can compute Keq for OH + Hg = HOHg
b) Use the results in (a) plus the forward rate constant to get the rate constant for HOHg dissociation
c) Show that HOHg-Y bond energies mimic BrHg-Y bond energies
d) use global modeling to show that, on a global basis, OH adds very little to oxidation of Hg(0)
2019
BrHgO• + C2H4 and BrHgO• + HCHO in Atmospheric Oxidation of Mercury: Determining Rate Constants of Reactions with Pre-Reactive Complexes and a Bifurcation. Khoa T. Lam, Curtis J. Wilhelmsen, and Theodore S. Dibble, J. Phys. Chem. A 2019, 123, 6045-55.
Computational Study on the Photolysis of BrHgONO and the Reactions of BrHgO• with CH4, C2H6, NO, and NO2: Implications for Formation of Hg(II) Compounds in the Atmosphere. Khoa T. Lam, Curtis J. Wilhelmsen, Abraham C. Schwid, Yuge Jiao, and Theodore S. Dibble, J. Phys. Chem. A 2019, 123, 1637-47.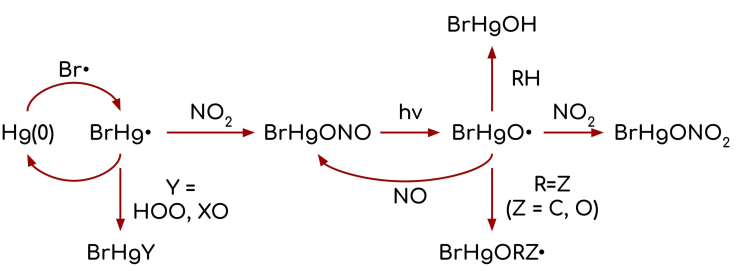
2018
Comment on “Isomerization of the methoxy radical revisited: the impact of water dimers” … and “Isomerization of methoxy radical in the troposphere: competition between acidic, neutral and basic catalysts” …. T. S. Dibble, Phys. Chem. Chem. Phys., 2018, 20, 11481-11482.
2017
Structures, Vibrational Frequencies, and Bond Energies of the BrHgOX and BrHgXO Species Formed in Atmospheric Mercury Depletion Events. Y. Jiao and T. S. Dibble, J. Phys. Chem A 2017, 17, 6353-6371.
A new mechanism for atmospheric mercury redox chemistry: implications for the global mercury budget. H. M. Horowitz, et al., Atmos. Chem. Phys. 2017, 17, 6353-6371.
First kinetic study of the atmospherically important reactions BrHg• + NO2 and BrHg• + HOO. Y. Jiao and T. S. Dibble, Phys. Chem. Chem. Phys. 2017, 19, 1826-1838.
2016
Thermodynamics limits the reactivity of BrHg• radical with volatile organic compounds. T. S. Dibble and A. C. Schwid, Chem. Phys. Lett. 2016, 659, 289-294.
Tunneling effect in 1,5 H-migration of a prototypical •OOQOOH. Y. Sha and T. S. Dibble, Chem. Phys. Lett. 2016, 646, 153-157.
Critical Review of Atmospheric Chemistry of Alkoxy Radicals. T. S. Dibble and J. Chai, in Advances in Chemistry of the Contemporary Atmosphere: Volume I, J. R. Barter, T. J. Wallington, and A. Steiner, Eds, World Scientific, 2016.
2015
Quality Structures, Vibrational Frequencies, and Thermochemistry of the Products of Reaction of BrHg• with NO2, HOO, ClO, BrO, and IO . Y. Jiao and T. S. Dibble, J. Phys. Chem. A 2015, 119, 10502–10510.
Quantum Chemical Study of Autoignition of Methyl Butanoate. Y. Jiao, F. Zhang, and T. S. Dibble, J. Phys. Chem. A 2015, 119, 7282–7292.
2014
Quantum Chemistry Guide to PTRMS Studies of As-Yet Undetected Products of the Bromine-Atom Initiated Oxidation of Gaseous Elemental Mercury. T. S. Dibble, M. J. Zelie, and Y. Jiao, J. Phys. Chem. A 2014, 118, 7847–7854.
Pressure Dependence and Kinetic Isotope Effects in the Absolute Rate Constant for Methoxy Radical Reacting with NO2. J. Chai and T. S. Dibble, Int. J. Chem. Kinet. 2014, 46, 501-511.
Rate Constants and Kinetic Isotope Effects for Methoxy Radical Reacting with NO2 and O2. J. Chai, H. Hu, T. S. Dibble, G. S. Tyndall, and J. J. Orlando. J. Phys. Chem. A 2014, 118, 3552–3563.
2013
Quantum Chemistry, Reaction Kinetics, and Tunneling Effects in the Reaction of Methoxy Radicals with O2. H. Hu and T. S. Dibble. J. Phys. Chem. A 2013, 117, 14230–42.
2012
Thermodynamics of reactions of ClHg and BrHg radicals with atmospherically abundant free radicals. T. S. Dibble, M. J. Zelie, and H. Mao. Atmos. Chem. Phys. 2012, 15, 17887-17911.
Cis-trans isomerization of chemically activated 1-methylallyl radical and fate of the resulting 2-buten-1-peroxy radical. T. S. Dibble, Y. Sha, W. F. Thornton, and F. Zhang. J. Phys. Chem. A 2012, 116, 7603–7614.
Temperature-Dependent Branching Ratios of Deuterated Methoxy Radicals (CH2DO•) Reacting With O2. H. Hu, T. S. Dibble, G. S. Tyndall, and J. J. Orlando. J. Phys. Chem. A 2012, 116, 6295–6302.
2011
Impact of tunneling on hydrogen-migration of the n-propylperoxy radical. F. Zhang and T. S. Dibble, Phys. Chem. Chem. Phys., 13, 17969-77.
Effects of Olefin Group and Its Position on the Kinetics for Intramolecular H-Shift and HO2 Elimination of Alkenyl Peroxy Radicals. F. Zhang and T. S. Dibble, J. Phys. Chem. A, 115, 655-63.
Understanding OH Yields in Electron Beam Irradiation of Humid N2. K. L. Schmitt and T. S. Dibble. Plasma Chem. Plasma Proc., 31, 41-50.
2010
Potential energy profiles for the N + HOCO reaction and products of the chemically activated reactions N + HOCO and H + HOCO . T. S. Dibble and Y. Zeng. Chemical Physics Letters, 495, 170-4.
Atmospheric chemistry of isopropyl formate and tert-butyl formate. A. S. Pimentel, G. S. Tyndall, J. J. Orlando, et al., International Journal of Chemical Kinetics, 42, 479-498.
2009
Towards a Consistent Chemical Kinetic Model of Electron Beam Irradiation of Humid Air. K. L. Schmitt, D. M. Murray, and T. S. Dibble. Plasma Chemistry and
Plasma Processing, 29, 347-362.
Optical diagnostics of a low power—low gas flow rates atmospheric-pressure argon plasma created by a microwave plasma torch. C. Wang, N. Srivastava, S. Scherrer,
P.-R. Jang, T. S. Dibble and Y. Duan. Plasma Sources Science and Technology, 18, 025030.
A study of OH radicals in an atmospheric AC discharge plasma using near infrared diode laser cavity ringdown spectroscopy combined with optical emission spectroscopy. N. Srivastava, C. Wang, and T.S. Dibble. European Journal
of Physics D, 54, 77-86.
Observation and quantification of OH radicals in the far downstream part of an atmospheric microwave plasma jet using cavity ringdown spectroscopy. C. Wang, N.
Srivastava, and T. S. Dibble. Applied Physics Letters, 95, 051501.
2008
Failures and limitations of quantum chemistry for two key problems in the atmospheric chemistry of peroxy radicals. T. S. Dibble. Atmos. Environ., 42, 5837-48.
2007
Computational Studies of Intramolecular Hydrogen Atom Transfers in the β-Hydroxyethylperoxy and β-Hydroxyethoxy Radicals. K. T. Kuwata, T. S. Dibble, E. Sliz, and E. B. Petersen. J. Phys. Chem. A, 111, 5032-42.